17 April 2021
Integrating Medical Device Product Development, Design for Six Sigma and Quality Systems Requirements – Part 1
Introduction
In today’s competitive, global environment, innovation and new product development are critical to a company’s growth and sustainability. New products that differentiate companies from their competitors serve as a competitive advantage and strategic strength.
Organic growth is the focus of many organizations that have spent the last few decades in intensive cost-cutting initiatives, processes and measures. A structured, flexible and effective new product development process that facilitates speed to market, reduces product development cycle times, manages risks, and delivers products that meet the requirements of customers and the marketplace enhances the probability of product launch success.
For the medical device industry, documenting new product development activities from concept, design and manufacturing, to sales and distribution in a controlled manner is detailed in the 21 CFR Part 820 Medical Devices – Quality System Regulation – Current Good Manufacturing Practices, and ISO 13485:2016 Medical Devices – Quality Management Systems – Requirements for Regulatory Purposes. Integrating the needs and requirements of the regulation and standard into a corporate product development process can improve efficiencies, minimize risks and enable compliance to FDA and ISO regulations.
Part 1 of this article describes a new product development process that incorporates Design for Six Sigma (DfSS) tools. Part 2 will demonstrate how DfSS enables an organization to integrate the medical device quality system requirements into the new product development process.
Best Practices for New Product Development
Dr Robert Cooper of MacMaster University, Canada, identified that only 11% of product development failures are for technical reasons associated with the performance of the product itself.1 The predominant causes of new product failure are marketing or market related. These causes include the development of products the customer did not want and the development of me-too products. Cooper also identified that the single most important factor in determining a new product’s potential success was the presence of clearly superior product features that were well differentiated, offered significant customer benefits, and fulfilled unmet customer or market needs.
In April 2002, Industry Week published a survey on new product development. See Table 12.
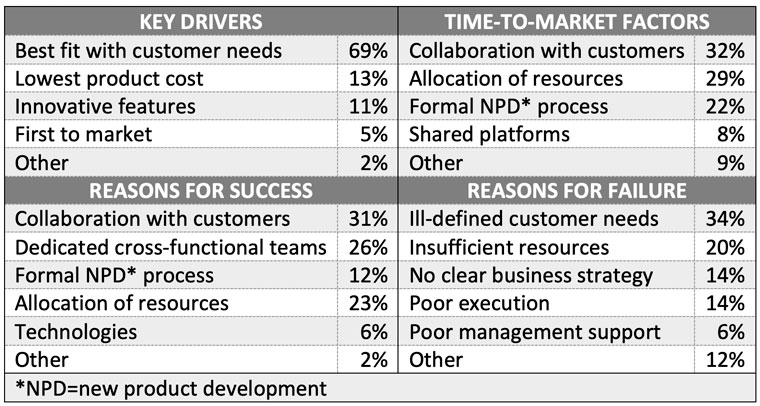
In a March 2005 survey, Pete Collins from PriceWaterhouseCoopers found that 78% of fast growth CEOs mentioned new product development as a top priority that fit with strategic and corporate values.3 Developing new products has improved company revenues, earnings and profit margins, and has fundamentally changed their business processes.
Based on these studies, manufacturers can infer that success in new product development and innovation depends on several factors. A recipe for a successful product, therefore, should include the following ingredients:
- Clearly defined customer and product requirements up front, as well as customer collaboration.
- Management buy-in, support, and involvement with a clear business strategy and a shared vision.
- Product development considered as a business process, not relegated to technology and engineering alone.
- Dedicated resources and cross-functional teams (involving marketing, technology, production, sales, regulatory, legal, and purchasing) all the way from concept through commercialization.
- Clear metrics, relevant data, and data-driven decisions.
- A structured, flexible, quality, product development process from concept to commercialization for the entire organization.
New Product Development Process
More than 70% of Fortune 500 companies have some sort of a formalized product development process.1 Product development processes range from three-stage processes to five- and six- stage processes (See Figure 1). It is imperative for companies to train all their employees across all functions about the process, so that the employees know what their roles are within it. Any business process is effective only when everyone shares the knowledge, and everyone is integrally involved in using it.

Simple three- and four-stage product development processes often bundle too many activities together in a single stage and trivialize the importance of vital but less-obvious activities. As a result, critical steps are missed, eliminated or overlooked. A new product development process is effective only if it includes all activities from the initial idea to final product sales.
The most commonly used process is the Stage Gate® Process first introduced by Dr. Robert Cooper in 19884. It consists of five activity stages, between each of which is a decision-point or a gate for a go/no-go decision.
Six Sigma
Six Sigma is a business process that guides companies to produce high quality products, reduce costs, improve efficiencies and increase profits of existing products, processes and services. Design for Six Sigma (DfSS) concepts can be included into an existing product development process or can be used to create a new product development process within an organization. Employing Six sSigma can also improve profits of existing products, processes and services. First introduced by Motorola, the system relies on using rigorous data to drive decision making. By implementing the Six Sigma process, many companies like General Electric, Honeywell, Raytheon and IBM have realized billions of dollars in cost savings, process improvement and quality improvement.
The goal of Six Sigma Is to produce products with extremely low defects — 3.4 defects in 1 million opportunities equivalent to a 99.99966% yield — based on those requirements that are critical to the customer. These requirements are called the critical-to-quality characteristics or CTQs. Figure 2 depicts a six-sigma capable process and displays the relationship between defects, yields and the sigma level or sigma capability. Thus, a six-sigma product or process has 3.4 defects in one million opportunities with a yield of 99.99966%, and a three-sigma product has 66,807 defects in one million opportunities, with a yield of 93.3%.
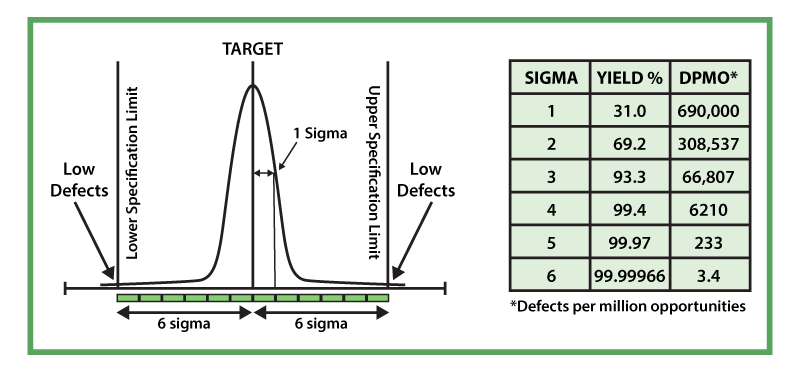
Defects can be quantified for any product, process, service or transaction. Rigorous root-cause analysis and statistical techniques can be used to identify the source(s) of the defects, and corrective actions can be taken to implement a solution(s) and ensure that the defect never occurs again. The Six Sigma process is a five-step process that has been widely used around the world. These steps are: Define > Measure > Analyze > Improve > Control
- Define customer requirements, the problem, the project scope, and the customer goals.
- Measure the current product or process capability.
- Analyze what is wrong and identify possible root causes and solutions.
- Improve the product or process by evaluating and testing the possible solutions.
- Control the product or process quality by implementing, monitoring, and sustaining the optimal solution.
Design for Six Sigma
The preamble to 21 CFR Part 820 Medical Devices – Quality System Regulation specifies that manufacturers “…cannot inspect quality into the product, [they] have to build it into the design and process.”5
By that token, device manufacturers can use DfSS as a tool for building quality into new products. DfSS is both a business management strategy and a product development process. It uses metrics, data and statistics, team dynamics, risk management, and project management tools to take products from concept to commercialization. The data-driven decision-making process delivers a six-sigma-capable product or service by focusing on up-front design and process parameters based on customer and market requirements.
DfSS incorporates the Six Sigma data-driven principles into the new product development process. New products are designed, developed and distributed to Six Sigma quality and capability for those critical to quality requirements specified by the customer, market, or intended use.
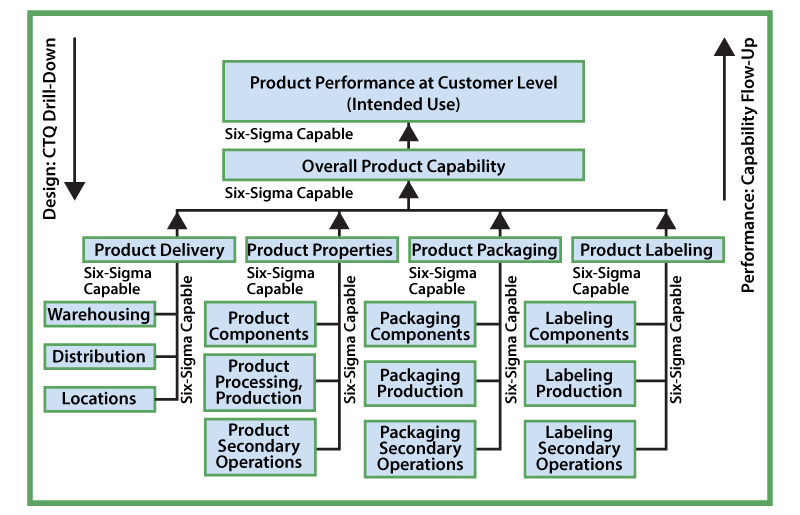
DfSS uses a systematic approach. Manufacturers drill down from the customer’s requirements to detailed product properties and specifications. From there they consider product architecture and design (based on product requirements), and finally the detailed process controls that must be employed during production to ensure that a product meets the customer’s requirements (Figure 3).
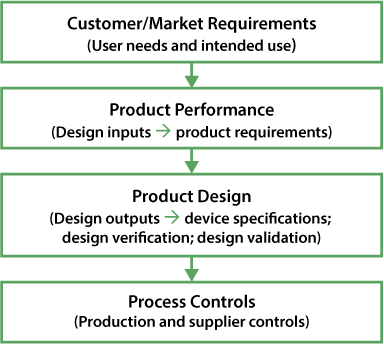
These quantifiable waterfall and drilldowns identify acceptance criteria, specifications and tolerances for the finished device or product and facilitate documentation to comply with the quality system requirements.
The Design for Six Sigma process provides a series of tools that are especially suited to medical device manufacturing. Such tools include:
- Portfolio management. Organizations can assess product portfolios and formulate short-, mid- and long-term growth strategies.
- Risk management. DfSS identifies and addresses the risk at the end of each phase, enabling the teams and management to make informed decisions. Such practices can minimize potential risks and failures during design, production, sales and end-use.
- Communication. Standardized processes and templates facilitate communication between the team, the business and the customers. DfSS is very effective in working with remote and virtual teams.
- Project management. The structured product development process provides a clear understanding of the processes, deliverables, activities, timelines and budgets.
DfSS is not just about designing the product. The goal is that the new product, process or service should have robust (six sigma) performance. These outcomes should be seen by the customer, consumer, or end-user. They should be visible during product launch, as well as during production. In short, the Six Sigma principles should be included in the product by design. An example of a six-step new product development process is shown in Figure 4. There are six activity phases and six decision-making points or tollgates. The nomenclature in the figure is an extension of the Six Sigma roadmap.
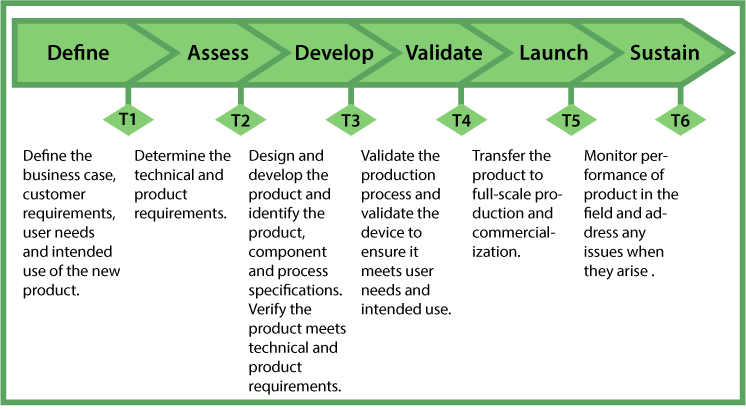
(T = Tollgate Review after each stage)
The DfSS process was originally used for the design and engineering of complex machines and parts, where the overall product’s performance is dependent upon all of its sub-assemblies and components. The design specifications and capability of every individual component and sub-assembly is quantified by a rigorous, step-by-step, drill-down from the finished product to every individual component. If each component and sub-assembly is six sigma capable within its specifications, it ensures six sigma capability of the final product (Figure 5). In the highly regulated medical device industry where performance and safety are critical, DfSS fits well for medical device new product development. In the medical device scenario, each new product is designed so as to ensure defect-free performance. In other words, it is designed for its intended use with six sigma capability.
Myths and Misconceptions of DfSS
One of the biggest myths of DfSS is that it introduces extraordinary amounts of bureaucracy to the new product development process. There is a perception that it increases timelines and delays product launches. In fact, the opposite is true. By identifying the characteristics that are critical to quality, DfSS focuses on the right scope, deliverables and the necessary activities of each stage providing speed and flexibility to the process. Teams are empowered to make decisions, but they are also held accountable for those decisions. Decisions are made by assessing and addressing all potential risks.
Another misconception is that a structured new product development process stifles creativity and innovation. DfSS actually stimulates innovation in product development by focusing on understanding the unmet needs of the market or the customer. Products that fulfill those unmet needs are the result of innovative solutions and execution.
Product development is all about bringing new products to market leveraging validated and proven technologies and core competencies. Long-term research that creates breakthrough and disruptive technologies should be conducted at a basic research level.
These activities should be part of the long-term growth plan of the organization. Once the capabilities and potential of the breakthrough technologies have been validated, they can be used as platforms to launch and spin off many new products for specific customers and applications using DfSS and new product development process.
The biggest hurdle for any initiative is the buy-in, support and involvement of upper management. There must be a shared vision from high-level managers. They must be integrally involved in the process and facilitate the cultural change. Results from such growth initiatives often take between two and three years to realize, compared to cost-cutting and productivity programs that realize benefits in six months to one year. The commitment must be sustained for the long and foreseeable future.
Benefits of Design for Six Sigma
The benefits of a rigorous, data-driven product development process include:
- Clearly defined product requirements based on intended use.
- A fundamental understanding of the components and processes used to manufacture the device.
- Differentiated and high-quality new products.
- Cycle time reduction and speed to market.
- Increased revenues and profits.
- Flexibility to respond to new requirements, applications and regulations.
- Better assessment of risks through the product development cycle.
- Sustainable products and processes.
- Effective investigations and root cause analysis if there are issues in production or in the field during use.
Conclusion:
DfSS can be integrated into a company’s existing product development process. It uses a systematic, data-driven approach to quantify critical product requirements based on the intended use. Products are designed and developed by fundamentally understanding how components, sub-assemblies and processing quantifiably relate to the performance of the finished device.
Finished devices can be designed, developed, manufactured, and sold with Six Sigma quality and capability, meeting the end-user needs for safe and effective products. Implementing the concept, leads to product innovation, consistency, and reliability, as well as sustainable sales, revenues and profits.
REFERENCES
- Robert G Cooper, Winning at New Products 3rd Ed, (New York City: Harper Collins, 2001)
- Richard Osborne, “New Product Development — Lesser Royals”, Industry Week 65, no 3 (April 2002): 251-253
- Pete Collins, Fast Growth Companies Make Innovation a Way of Life (New York City: PriceWaterhouseCoopers, March 2005)
- Robert G. Cooper “The New Product Process: A Decision Guide for Managers” Journal of Marketing Management 3, no 3 (1988): 238-255
- 21 CFR 820 Medical Devices – Quality System Regulation – Current Good Manufacturing Practices